
Chapter 2: Solvent Based Extraction (Part 1: Hydrocarbon Extraction)
, by Avery Benitez, 52 min reading time

, by Avery Benitez, 52 min reading time

INTRODUCTION TO SOLVENT-BASED BOTANICAL EXTRACTION

Botanical extraction is a fascinating process used widely to create some of the modern products used daily. From medicine to cleaning agents, powerful botanical compounds can be extracted from a wide variety of different plants and used to make a plethora of useful products.
Currently, there are several popularized methods of botanical extraction in today's extraction landscape, all of which have their own use cases and unique benefits. Most botanical extraction methods can be categorized into two different methodologies, either solvent-based extraction or solventless-based extraction.
In some cases, target compounds can be concentrated from biomass material utilizing solventless-based extraction techniques centered around mechanical separation methods including Ice water extraction, dry sifting, and Rosin pressing. These methods of extraction are limited in their utility and typically only apply to plants that contain their target compounds within glandular trichomes.
Conversely, solvent-based botanical extraction methods utilize organic solvents to dissolve target compounds from botanical biomass to form a solution. Solvent-based extraction methods have a wider array of applicable use cases and allow for greater versatility in an application when it comes to botanical extract. In this course, we will focus on the two most prominent and applicable forms of solvent-based extraction:hydrocarbon extraction and ethanol extraction.
While both methods are highly efficient in extracting botanical compounds, both have their own benefits and drawbacks. When it comes to determining the suitable form of solvent-based extraction for a specific process, it comes down to determining which solvent is most compatible with the target compounds polarity and boiling points, along which process fits the tolerance for potential hazard. This section will explore the basics of how solvent-based botanical extraction works and highlight the factors in determining which solvent is best for a specific application.
POLARITY
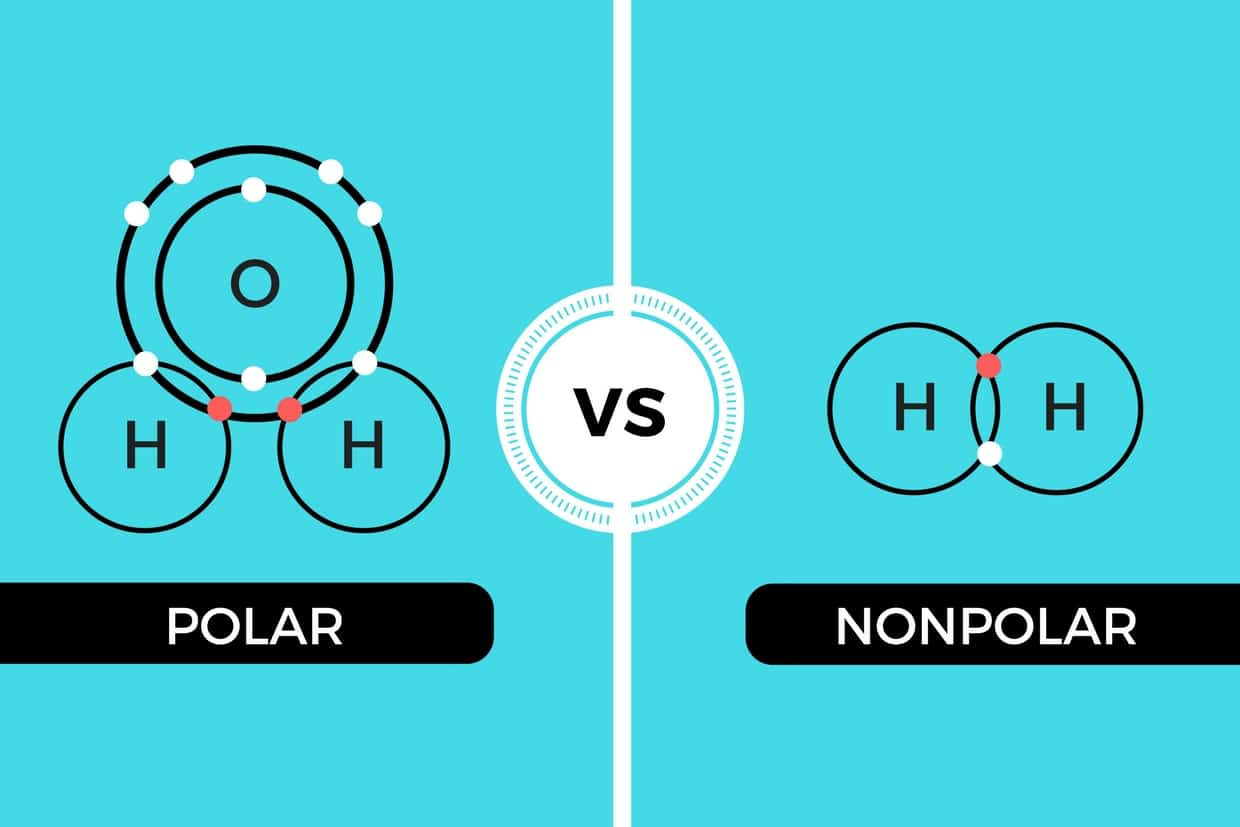
In the world of solvents like, dissolves like. A solvent’s polarity dictates a solvent’s capability to dissolve a target compound from biomass material. Generally, polar solvents can dissolve polar compounds, and nonpolar solvents can dissolve nonpolar compounds. When choosing an extraction solvent, it must be compatible with the target compounds’ polarity to dissolve them from the botanical plant matter to form a solution.
Solvent-based extractions rely on the principle of solubility in order to extract target compounds from botanical biomass. Solubility refers to a compounds' ability to be dissolved by a solvent forming a solution. A solution is defined as a homogeneous mixture of two or more substances.
The solvent is the major part of the solution in botanical extraction. The substance dissolved in the solvent, referred to as the solute, is the minor part of the solution. When a solution has dissolved all it can and can no longer dissolve any more solute, it is considered a saturated solution.
The compound’s solubility and saturation capacity within a solvent can be altered by increasing or decreasing the temperature of the solvent and or solution. However, the solubility of a compound within a solvent is ultimately determined by its polarity.
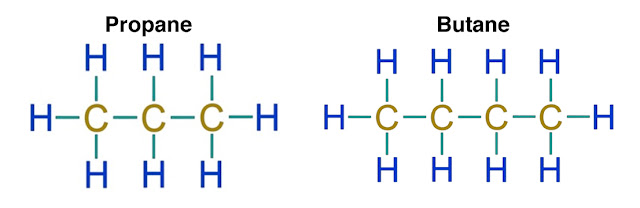
A substance’s polarity is determined by the distribution of electron density across the molecular structure of that compound. Compounds that have an even spread of electrons like butane and propane are nonpolar. Compounds with an unevenly shared electron density like water are classified as polar compounds.
Polar compounds are defined as having both a positive and a negatively charged end. The polarity of the solvent will determine the compounds it can dissolve. While some compounds can be more polar or nonpolar than others, generally like dissolves like, polar solvents can dissolve polar compounds, and nonpolar solvents can dissolve nonpolar compounds.
We can calculate a compound's polarity based on its electronegativity. Electronegativity measures how strongly an atom is attracted to an electron it bonds with. The higher this number, the more an atom of that element will be attracted to electrons in a bond.
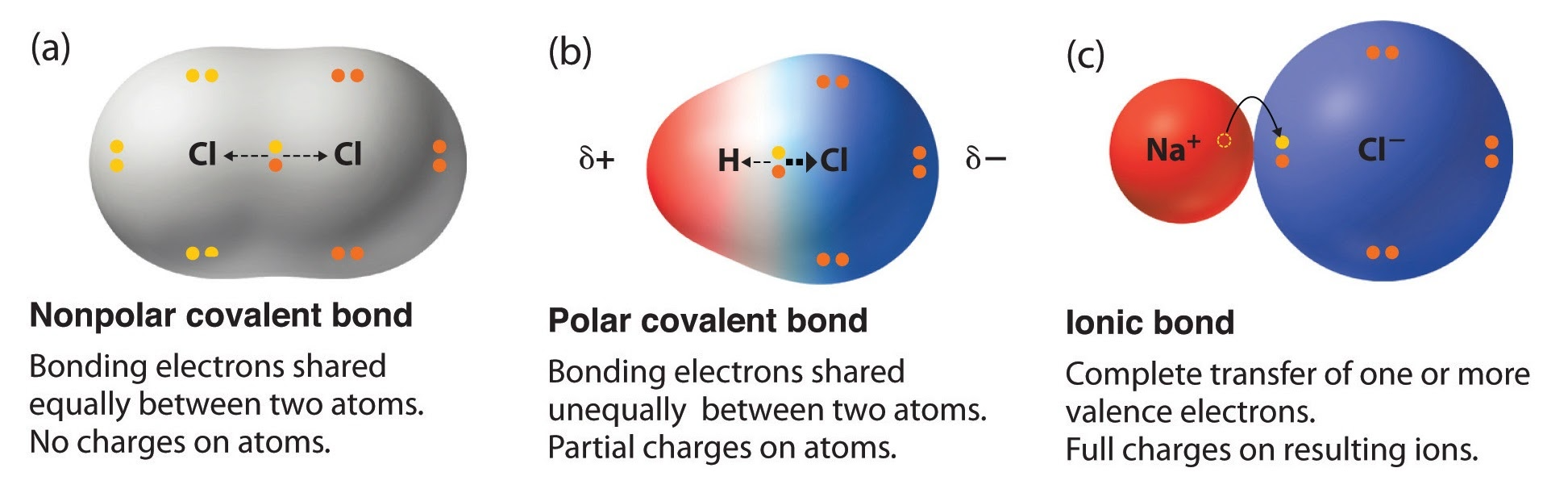
Generally, if the compound has an electronegativity <0.4, it is classified as a nonpolar covalent bond. If the compound’s electronegativity is between 0.4 and 1.8, it is considered a polar covalent (Polar), and if the electronegativity is greater than 1.8, it is considered an Ionic bond which is nonpolar.
While polarity is typically binary, meaning it is either polar or nonpolar, there are some instances where a compound has both polar and nonpolar properties, like in the case of ethanol. Ethanol's polarity is unique, while its electronegativity classifies it as polar. Due to its structure, ethanol has a hydrotropic polar head(water-loving) that can bind to water-soluble polar compounds and a nonpolar tail that can dissolve hydrophobic or oil-soluble nonpolar compounds that are typically found in botanical biomass. When a compound has both polar and nonpolar properties, it is classified as amphiphilic, meaning it has both hydrophilic and hydrophobic properties.
ETHANOL

Ethanols' amphiphilic nature can be beneficial when making a full spectrum extract containing all the different compounds found within the botanical biomass. However, it can make the processes of botanical extract refinement more challenging when trying to make a highly concentrated botanical extract of a certain compound.
While a solvent’s polarity plays a major role in its ability to dissolve both desirable and undesirable compounds from botanical biomass, the solubility and saturation capacity of a solvent can be altered to some extent by adjusting the temperature of the solvent prior to extraction—generally, both the solubility and saturation capacity of a solvent increases or decreases along with its temperature.
As the temperature of a solution increases, typically, its solubility and saturation capacity also increases. When the temperature of a solvent is decreased, its solubility and saturation capacity also decreases. In botanical extraction, it is quite common for the solvent to be chilled prior to extraction to alter its selectivity and limit the co-extraction of undesirable compounds.
DC-40 Inline Ethanol Chiller by Delta Separations
Chilling solvent prior to extraction reduces both the solubility target compounds and undesirable plant waxes and chlorophyll. While this is somewhat counterintuitive, chilled can create a higher purity botanical extract. While the temperature of a solvent plays a role in the solubility and saturation capacity of target compounds within a solvent, ultimately, a compound’s solubility within a solvent is predominantly determined by its polarity.
When it comes to botanical extraction, compatibility between a solvent's polarity and the target compounds' polarity is essential in extracting the target compounds from botanical biomass. While polarity is quite possibly the most important factor in extracting the target compounds, the solvents boiling point in relation to that of the target compounds will determine the viability of recovering that solvent from the botanical extract solution.
BOILING POINT
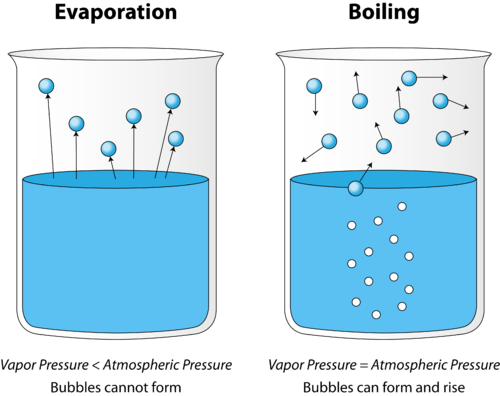
A compound’s boiling point is defined as the temperature at which the vapor pressure of a liquid equals the atmospheric pressure of the surrounding liquid and phase changes from a liquid to a vapor observed as boiling. When it comes to a solvent-based extraction, the solvent’s boiling point in relation to the target compounds' boiling point will determine the viability of recovering said solvent from the botanical extract without evaporating the target compounds.
A solvent with a lower boiling point than that of the target compounds is ideal to evaporate the solvent from the botanical extract solution effectively. Ideally, there should be a significant difference between the solvent's boiling point and the target compound's boiling points so the solvent can be easily evaporated from the solution without co-evaporating the target compounds.
For instance, if the target compound is cannabidiol, most extraction solvents have boiling points that are well below the boiling points of cannabidiol (180C/356F), making them easily recoverable from the botanical extract with limited degradation of the target compound, making them considerable candidates. However, higher boiling point solvents like ethanol would not be strong candidates if the target compounds are highly volatile, like terpenes.
50L Neocision Rotary Evaporator Turnkey System By BVV
In this case, recovering the high boiling point solvent from the extract would result in the volatilization or conversion of the target terpenes making it nearly impossible to separate the two through distillation. In this case, a nonpolar solvent with a lower boiling point like butane or propane would be a better candidate to extract lower boiling point terpenes.
When choosing the right solvent to extract target compounds the extraction solvent should have a lower boiling point with a great enough difference between the extraction solvent and the target compounds boiling points. This will allow the solvent to be easily recovered from the extract without co-evaporating the target compounds in order for it to be considered a viable method of extraction.
Once a solvent has been determined to be compatible with the target compounds’ polarity and boiling points, the final key factor determining the overall viability of a solvent-based botanical extraction method for application is its potential hazard. Inherent risks of the extraction method must be considered, the appropriate fire safety measures required, the overall suitability of the facility, and potential zoning or licensing restrictions to determine if an extraction method is viable for the application.
SAFETY

When performing a solvent-based extraction, safety should always be the number one concern. Typically most solvents used for botanical extraction present an inherent risk due to their flammability and volatility. Although one solvent may be a prime candidate to extract target compounds, the level of hazard or fire safety requirements may disqualify that method based on the criteria.
While some extraction methods pose more inherent risk than others with the right equipment, solvent-based extractions can be performed with very little risk posed to the operator or the surrounding area. When performing a solvent-based extraction with flammable and volatile solvents, the best way to mitigate risk is to perform solvent-based extraction methods within an appropriately rated hazardous location.
Utilizing highly flammable solvents under extreme temperatures and pressures can be dangerous. To safely and compliantly perform most forms of solvent-based extraction, it must be done in an appropriately rated hazardous location with all the proper safety and protective equipment.
Hazardous locations are areas where the possibility of fire or explosion hazards may exist under normal or abnormal conditions because of the presence of flammable, combustible, or ignitable gases, vapors, liquids, dust, or fibers/flyings. These areas are classified solely for the purpose of ensuring the safe and proper specification and installation of electrical/electronic equipment. Using an appropriately rated extraction booth or fume hood greatly reduces the risks of working with volatile organic solvents.
C1D1 Explosion-Proof Booth By Advanced Extraction Labs
Hazardous locations fall under three classifications with two separate divisions. Class 1 locations are those in which flammable vapors may be present, Class 2 locations are those in which combustible dust may be found, and class 3 locations are those in which easily ignitable fibers or flyings may be present. Most solvent-based botanical extract processing techniques fall under the first classification due to the possibility of flammable vapors being present.
Depending on the specific botanical extract processing technique it may be classified as division 1 or division 2; these divisions define the likelihood of hazardous materials in a flammable concentration. In a division 1 environment, ignitable concentrations of hazards exist under normal operating conditions or where a hazard is caused by frequent maintenance, repair work, or equipment failure. Division 2 environments are defined as environments where ignitable concentrations of hazards are handled, processed, or used, while normally in closed containers or closed systems from which they can only escape through accidental rupture or breakdown of such containers or systems.
Lower Explosive Limit Gas Detector By Honeywell
Hazardous locations require all electrical/electronics equipment to be designed, tested, and labeled as acceptable for use in the areas where it is installed. Beyond utilizing appropriately rated electrical/electronic components most extraction areas will come equipped with an explosion-proof exhaust fan providing a high air exchange rate, along with a lower explosive limit gas detector to alert high levels of gas and trigger an increased rate of exhaust ventilation.
An appropriately rated extraction booth is a must-have to safely and compliantly perform solvent-based extraction. It is also required by most local municipalities and therefore should be worked into the budget of every botanical extraction lab build. Keep in mind that some of the more volatile extraction solvents like hydrocarbons typically require operation in a C1D1 environment or may not be allowed within the specific zoning or licensing. It is important to always consult with the local fire marshal to determine the viability and legality of performing certain types of solvent-based extraction methods before building out a solvent-based extraction lab.
By consulting with a local fire marshal, the exact requirements necessary to perform solvent-based extraction methods and store solvent in the facility can be determined. While there are some broad requirements to follow, the local fire marshal will determine the specific requirements for each facility. Now that we have gone over some of the differentiating factors between solvent-based extraction methods, let’s explore the solvent-based extraction methods covered in this chapter.
HYDROCARBON EXTRACTION
10LB+ EVO Certified Closed Loop Extractor by BVV
Hydrocarbon extraction is an incredibly versatile method of botanical extraction. Hydrocarbon solvents like butane and propane are praised for their relatively high yield and low cost. Hydrocarbons are a classification of organic compounds composed of hydrogen and carbon. These highly volatile compounds are valued for their low boiling points, making them easily recovered from most botanical extracts.
Both butane and propane are nonpolar, making them great candidates to extract nonpolar botanical compounds. Butane and propane are gaseous at room temperature, and ambient pressure becomes liquid when pressure is increased, or temperature is reduced. Using liquid hydrocarbon solvent to saturate biomass material can dissolve target nonpolar compounds and extract them from the plant material. Due to butane and propane's lower boiling points, they lend themselves to preserving highly volatile compounds like terpenes resulting in a more aromatic and flavorful botanical extract.
Hydrocarbon extraction is an excellent option for creating high-quality extracts with decent throughput and relatively low startup cost in comparison to the level of equipment needed to create an ethanol extracted concentrate of similar potency. Although there are many benefits to Hydrocarbon extraction, the main drawback of Hydrocarbon extraction is the solvent's high volatility; hydrocarbons are highly combustible, requiring careful safety measures, including operation within a C1D1 extraction booth. Due to hydrocarbons’ high volatility, this extractionmethod may not be permitted by local municipalities. Be sure to consult with the local fire marshal to determine the viability of this method for zoning or licensing.
ETHANOL EXTRACTION
45L Jacketed Stainless Steel centrifuge By BVV
Ethanol extraction is an excellent method of botanical extraction valued for its high throughput potential and reduced flammability compared to more volatile solvent-based extraction methods like hydrocarbon extraction. Ethanol is classified as alcohol made from starch-based plant materials through a complex fermentation and distillation process. Ethanol (EtOH), commonly known as ethyl alcohol or grain alcohol, is a colorless flammable liquid with an incredible number of uses beyond botanical extraction including fuel and cleaning agent.
As an extraction solvent ethanol is unique due to its amphiphilic nature meaning it has both polar and nonpolar properties. Ethanol has a hydrotropic polar head(water-loving) that can bind to water-soluble compounds and a nonpolar tail that can dissolve hydrophobic or oil-soluble compounds typically found in botanical biomass. As the plant matter is saturated with ethanol, it dissolves both polar and nonpolar compounds from the biomass material extracting them. Ethanol's amphiphilic nature can be ideal when seeking to create a more full-spectrum product, but it can be less than ideal for those looking to create a more concentrated extract.
Due to ethanols' polar and nonpolar nature, it has an increased propensity to co-extract undesirable plant waxes and chlorophyll during the extraction process. This typically results in a less potent and pure end product requiring more post-processing to reach the same purity level than that of a nonpolar solvent like butane or propane. While ethanols’ selectivity for undesirable plant waxes and chlorophyll can be altered by chilling the ethanol prior to extraction, reducing the solubility of undesirable plant waxes and color pigments, it also reduces the solubility of target compounds.
Beyond its polarity, ethanol has a high saturation capacity and only requires a short retention time to dissolve target compounds lending itself to short runtimes and high throughput potential. Ethanol is particularly well suited for bulk processing, with available systems that can extract thousands of pounds of biomass within a single working shift. This makes ethanol a great option for those making botanical extracts at larger scales. Additionally, ethanol is less volatile than hydrocarbons, typically only requiring operation in a C1D2 hazardous location.

Chapter 2: Solvent Based Extraction Glossary
Azeotrope: A mixture of 2 or more liquids, which has a constant shared boiling point and vapor composition through distillation.
HYDROCARBON EXTRACTION
10LB+ EVO Certified Closed Loop Extractor by BVV
Hydrocarbon extraction is a widely used botanical extraction method that utilizes light hydrocarbon solvents like butane and propane to extract botanical compounds. Hydrocarbon extraction is preferred by many botanical extractors due to its high yields and low solvent recovery temperatures. These highly versatile extraction solvents lend themselves to producing a high purity botanical concentrate with little to no post-processing. Hydrocarbons are a classification of organic compounds made from carbon and hydrogen. Hydrocarbons are formed due to the compression of animal and plant remains over long periods and are pulled from porous rocks where they pool and concentrate. These alkane molecules have an even distribution of electrons, making them nonpolar and well suited for extracting nonpolar botanical compounds.
The most commonly used hydrocarbons for botanical extraction include butane and propane. These hydrocarbons can be utilized on their own to perform botanical extraction, or they can be combined into a blend of hydrocarbon solvents and utilized to extract a fuller spectrum of botanical compounds from biomass material. Typically a blend of n-butane, Isobutane, and propane is utilized to create an extraction solvent with greater affinity to extract a wider variety of compounds found within botanical biomass resulting in a more full-spectrum extract.
120LB DIPTUBE Tank High Purity USA 70/30% N-Butane/Propane Blend By BVV
Both butane and propane are class 1 group D flammable liquified gases that the Food and Drug Administration (FDA) have generally recognized as safe (GRAS). These light hydrocarbons are gaseous at room temperature and ambient pressure where they are denser than air and tend to pool near the floor. When pressurized or placed under reduced temperature they become liquified and can saturate biomass material and extract nonpolar botanical compounds.
20LB High Purity N-Butane By BVV
Hydrocarbons are the preferred extraction solvent of many extractors looking to preserve highly volatile aromatic compounds due to their low boiling point -1C/30.2F for butane and -42C/-43.6F for propane. Since these solvents boil off at a lower temperature than most aromatic compounds, they can be recovered with minimal heat preserving more aromatic compounds in comparison to other high boiling point extraction solvents like ethanol. While butane and propane's low boiling points lend themselves to aromatic compound preservation, it also makes hydrocarbons highly volatile and more hazardous to work with.
Although butane is generally recognized as safe by the FDA and can be consumed by humans in trace amounts, utilizing these highly volatile and flammable hydrocarbons as organic solvents under extreme temperatures and pressures can be dangerous. While it is highly unlikely that hydrocarbon extraction will result in an explosion when performed properly, when utilizing these highly flammable gases, there is always an inherent potential ignition. For this reason, any and all potential sources of heat or flame should be kept far away from areas in which hydrocarbon gases are present. To further mitigate the potential for ignition, hydrocarbon extraction should be performed inside of a C1D1 extraction booth with all the proper safety and protective equipment.C1D1 Explosion-Proof Booth By Advanced Extraction Labs
Among the most important pieces of safety equipment for hydrocarbon extraction is a C1D1 extraction booth which greatly reduces the potential hazard when working with hydrocarbon solvents. A C1D1 hazardous location is where ignitable vapor concentrations exist under normal operating conditions and where a hazard is caused by frequent maintenance, repair work, or equipment failure. Hydrocarbon extraction typically falls under a Class 1 Division 1 environment because ignitable concentrations of hydrocarbons exist under normal operating conditions. Depending on a specific municipality, the fire inspector may also require said extraction booth to have a fireproof burn rating which is commonly required when dealing with larger volumes of solvent within the enclosed area.
C1D1 Hazardous locations require all electrical/electronics equipment to be designed, tested, and labeled acceptable for use in C1D1 areas. Beyond utilizing appropriately rated electrical/electronic components, most extraction areas will come equipped with an explosion-proof exhaust fan providing a high air exchange rate, along with a lower explosive limit gas detector to alert high levels of gas and trigger an increased rate of exhaust ventilation.
Lower Explosive Limit Gas Detector By Honeywell
While the use of appropriately rated electrical components within the extraction area drastically lowers the potential of a flammable substance being ignited, one of the more important functions of a C1D1 extraction area is to reduce the risk of an explosion by regulating the airflow of the enclosed space to keep the levels of gases below the lower explosive limit of the used gas.
A C1D1 extraction booth helps mitigate the risk of an explosion through the use of an explosion-proof exhaust fan, providing a high rate of air exchange. It comes with a lower explosive limit gas detector used to alert the user of high levels of gas and trigger an increased rate of exhaust ventilation. When extracting with hydrocarbons, it's best to keep the percentage of solvent vapors within the enclosure well below 25% of their lower explosive limit which is 1.86% for butane and 2.1% propane.
Portable Butane Leak Detector By BVV
When performing a hydrocarbon extraction inside a C1D1 rated Explosion-proof enclosure, only appropriately rated explosion-proof electronics should be utilized, and great care should be taken to limit exposure to open flame or static electricity by bonding metal containers during flammable solvent transfer and grounding equipment. A C1D1 Environment is a must-have to safely and compliantly perform hydrocarbon extraction and should be worked into the budget of every new hydrocarbon extraction lab build. While an explosion-proof environment significantly reduces risk during hydrocarbon extraction, to ensure personal safety while performing hydrocarbon extraction, personal protective equipment in the form of safety goggles, nitrile gloves, antistatic clothing, and a respirator should be worn at all times.
When operating within a C1D1 environment hydrocarbon extraction is typically performed utilizing a closed-loop hydrocarbon extractor. A closed-loop system allows for optimal safety as it contains all flammable solvents from the extraction process within a closed system and allows for the recapturing and reuse of solvent.
1LB MK-V Orthrus Bidirectional Flow Closed Loop Extractor By BVV
A typical closed-loop system can be broken down into three basic elements; The solvent tank/ recovery tank where the hydrocarbon blend is stored and recondensed, the material column where the plant matter is loaded and saturated, and the collection vessel where the botanical extract solution is collected and the solvent evaporation process is performed. While these three components are essential to the operation of a closed-loop system, several additional components can be utilized to improve the efficiency of a closed-loop system.
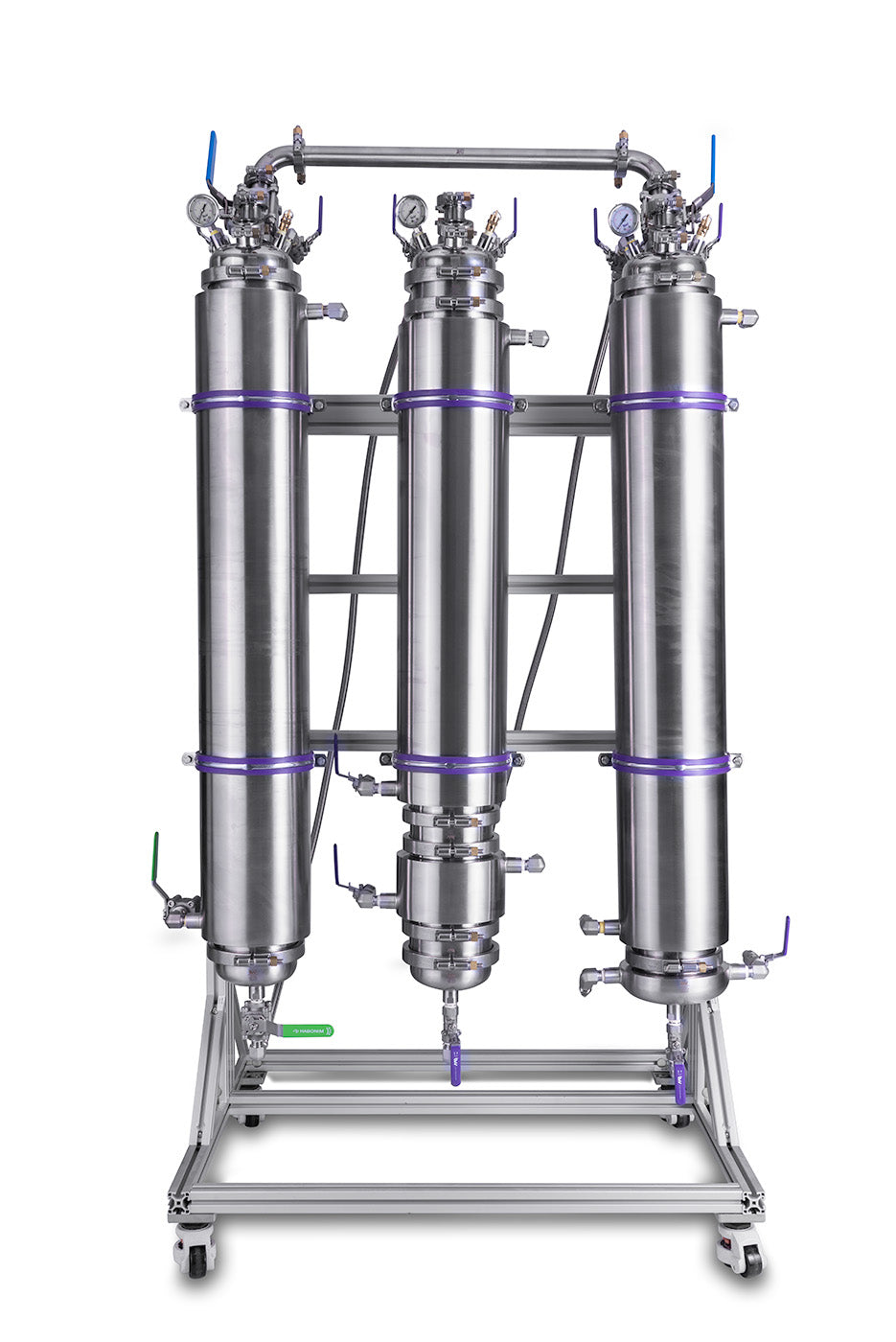
5LB EVO Certified Closed Loop Extractor *49 State Certified By BVV
Common optional additions to a closed-loop system include a dewax column used to precipitate and filter plant waxes from the solution, a color remediation column used to filter undesirable color pigments from the extract, and a molecular sieve column used to absorb any water from the solvent during the solvent recovery process. While the function of a dewaxing column can be substituted by chilling the extraction solvent prior to extraction and limiting its retention time, a molecular sieve and color remediation column are highly recommended to improve the overall efficiency of the extraction process. A color remediation column specifically is capable of drastically increasing the purity of the botanical extract and will be covered in great detail in the coming chapters.
2.5" Inline Filter Housing for Disposable Color Remediation Cartridges By AFS
Hydrocarbon extraction is performed simply by saturating the biomass material with a liquid hydrocarbon solvent. As the liquid solvent saturates the biomass, it dissolves compounds from the plant material. While this process is rather straightforward, several variables to the initial saturation can be optimized to increase the overall yield and purity of the resulting extract. During the initial saturation, the most effective variables to optimize the efficiency of a hydrocarbon extraction are solvent temperature, solvent to biomass ratio, and retention time.
Evo Elite Sleeved Solvent Tank By BVV
Solvent temperature is commonly altered either to increase the purity or overall yield of the hydrocarbon extraction. The extractor may choose to either chill the solvent to limit the pickup of impurities or extract using a room temperature or "warm" solvent to increase the overall yield of the extract. During extraction, the temperature of a solvent plays a big role in the solubility and saturation capacity of a solvent. Saturation capacity and solubility generally increase or decrease along with the temperature of the solvent.
When a solvent is chilled prior to extraction, its overall solubility decreases, which helps limit the co-extraction of undesirable impurities such as plant waxes, lipids, and chlorophyll. This improved selectivity is a key advantage of cold extraction processes.However, the reduced extraction efficiency observed at lower temperatures is not solely a chemical solubility phenomenon. It is also driven by physical changes in the solvent itself.
As temperature decreases, solvent viscosity increases, causing the solvent to behave in a thicker, more honey-like manner. This increased viscosity reduces mass transfer efficiency by limiting the solvent’s ability to readily penetrate the biomass matrix, diffuse through cellular structures, and fully solvate target compounds.
As a result, target compound pickup can be partially suppressed—not because the compounds are insoluble, but because the solvent is physically less able to access and mobilize them. This effect can be compensated for by increasing the solvent-to-biomass ratio, extending contact or retention time, or optimizing flow dynamics to restore adequate mass transfer while preserving the benefits of reduced impurity pickup.

Unistat 825 Dynamic Temperature Control / Circulation Thermostat By Huber
Hydrocarbon solvent is typically chilled within a -20C to -80C (-4F to -112F) range, increasing the selectivity of the extraction and limiting the co-extraction of impurities. Common methods of chilling hydrocarbon solvent include an injection coil submerged in dry ice that the hydrocarbon solvent runs through prior to injection of the material column or a jacked solvent tank connected to a refrigerated circulator to chill the hydrocarbon solvent prior to the initial saturation.
Stainless Steel Injection Coil By BVV
Conversely, a room temperature or "warm" hydrocarbon solvent can be utilized to perform the initial saturation. When utilizing a room temperature or "warm" hydrocarbon extraction, the solvent has greater solubility and saturation capacity allowing the solvent to more effectively dissolve target compounds from the plant matter. While this strategy allows for the target compounds to be more effectively dissolved from the plant matter, it also allows undesirable botanical compounds like color pigments and plant waxes to be more easily dissolved, resulting in a greater number of total impurities. Commonly, a warm extraction is paired with inline adsorbent filtration utilizing a color remediation column allowing for efficient extraction of the target compounds during the initial saturation and removing undesired impurities through filtering resulting in high yield and purity.
Color Remediation Column By BVV
Regardless of the extraction temperature strategy, once the solvent has reached the desired extraction temperature, the solvent is then injected into the material column saturating the biomass material. There are two methodologies for saturating the biomass material. Closing the outlet to the material column allows the material column to fill with solvent and fully saturate the plant matter increasing retention time before draining the solvent into the collection vessel. Another way is to leave the outlet of the material column open, allowing the solvent to quickly pass through the material column into the collection vessel, limiting solvent retention time for what is referred to as a quick wash. Retention time is an important factor in allowing the solvent to dissolve the target compounds. Too little retention time can result in reduced yield of target compounds, while too long retention time can allow more undesirable compounds like chlorophyll and plant waxes to be dissolved.

Icarus 5LB True Dewax Bidirectional Closed Loop Extractor (Discontinued) By BVV
Extended retention times are great for extracting everything from the plant matter. When this strategy is paired with inline adsorbent filtration, it can result in both high yield and purity. Conversely, a short retention time allows for greater purity by minimizing the time the solvent has to co-extract undesirables. When a quick wash/short retention time strategy is utilized in conjunction with chilled solvent, it can result in a concentrate with very low levels of impurities. Utilizing a short retention time and chilled solvent typically results in lower extract yield. This can be combated by utilizing a higher solvent to biomass ratio to ensure the majority of the target compounds are extracted while limiting the pickup of undesirable color pigments and plant waxes. Typically, a solvent to biomass ratio of 3-5:1 is commonly utilized to perform a hydrocarbon extraction. Up to a 10:1 ratio can be utilized when extracting with cold solvent to ensure the majority of the desired compounds are extracted.
20LB High Purity N-Butane By BVV
The solvent to biomass ratio is the final factor in the initial saturation that can be optimized. Most commonly, a ratio between 3-5 pounds of hydrocarbon solvent will be utilized for every 1 lb of biomass loaded into the material column. The greater the total volume of solvent utilized during the initial saturation, the greater the extraction efficiency. More solvent used for the initial saturation typically allows for more of the target compounds to be dissolved from the plant matter. An increased solvent to biomass ratio is especially useful when utilizing a quick wash, short retention time saturation strategy. While this ratio can be further increased to ensure the target compounds are extracted from the plant matter it results in a greater total volume of solvent used, increasing production costs, extending the solvent recovery process, and increasing overall runtime.

19 Liter Neocision Heated Circulator - ETL Listed By BVV
After the initial saturation is complete and the entirety of the botanical extract-rich hydrocarbon solution has been drained into the collection vessel, the bulk of the solvent recovery process is performed by heating the solution to evaporate the hydrocarbon solvent from the botanical extract solution. The solvent recovery process is a distillation process initiated by applying heat to the extract solution to evaporate the extraction solvent from the botanical extract and the condensation of the ensuing solvent vapors inside a chilled recovery tank. Typically the solution is heated by either submerging the collection vessel in a hot water bath or recirculating hot water through the jacket of the collection base.
4 Cylinder Butane Recovery Pump By BVV
The solvent recovery process can be performed utilizing two separate methodologies: active solvent recovery or passive solvent recovery. Both passive and active solvent recovery utilize heat applied to the collection to evaporate the solvent from the extract and cool the recovery tank to condense the hydrocarbon vapors inside a vessel separate from the extract. Active recovery assists the solvent recovery process by including a recovery pump that aids in the transfer and compression of the gaseous solvent, speeding up the solvent recovery process. While both methodologies have their benefits and drawbacks, both can be utilized to recover hydrocarbon solvents effectively.
MVP150 Explosion Proof Recovery Pump By Master Vapor Pumps
Regardless of the solvent recovery methodology, the collection vessel containing the extract solution is heated to initiate the solvent recovery process. The recovery tank is chilled and commonly pulled under vacuum only if the solvent tank is empty to create pressure and temperature differential between the heated collection base and the chilled recovery tank. This pressure and temperature differential between the two vessels results in the solvent vapors being attracted to the lower pressure and temperature recovery tank once the process connections between the two are opened.
DOT-Approved Recovery Cylinders By Mastercool
Note that if the recovery tank contains solvent, do not attempt to pull the recovery tank under vacuum as solvent vapors can be pulled out of the vessel into the vacuum pump, potentially causing ignition of the solvent vapor. While reduced pressure within the recovery tank assists in the travel of vapor from the collection vessel to the recovery tank, if the recovery tank still has solvent within it, the solvent recovery process can still be performed relying solely on the temperature differential between the two vessels. However, the pressure from the collection vessel must be greater than the pressure within the recovery tank, which can be ensured by keeping the recovery tank as cold as possible during the recovery procedure.

BVV-80C Chiller By BVV
During the solvent recovery process, the movement of the solvent vapor within the system operates under the principle that gas always seeks the lowest pressure region of a system. The pressure of a hydrocarbon gas directly correlates to its temperature by heating the hydrocarbon solvent within the collection tank, thus increasing its pressure. Moreover, by cooling the solvent recovery tank the ensuing vapors naturally are pulled towards the lower pressure chilled recovery tank where they are condensed and their pressure is reduced.
Neocision 5 Liter Heated Circulator - ETL Listed By BVV
The efficiency of the solvent recovery process is a function of the heat applied to the collection vessel and the amount of cooling applied to the solvent tank. Since hydrocarbon solvents have relatively low boiling points, light heat is sufficient to perform the evaporative process. While increased heat can help speed up the solvent recovery process, it is a double-edged sword as it may degrade or evaporate the more volatile compounds typically found in the botanical extract like terpenes. During this process, it is important to keep in mind the lowest boiling point of the compounds to be preserved, and stay under that temperature when determining the heat level to apply to the solvent recovery process.
Stainless Steel Condensing Coil By BVV
The other side of this solvent distillation process is the condensation of solvent vapors inside the recovery tank. Condensation happens when vapor is cooled or condensed past its condensation point and is initiated by the molecular clustering of vapor within a gaseous volume or at the contact of vapor with a cool enough liquid or surface. This process can be assisted through the use of a condensing coil submerged in dry ice, which provides a high surface area low-temperature zone where vapors can be condensed before entering the recovery tank. Typically the colder the recovery tank or condensing coil, the more efficiently the solvent will be recovered and collected.

7.5CF Neocision Vacuum Oven - ETL Certified By BVV
While it is typical for the bulk of the hydrocarbon solvent to be recovered during this time, depending on the specific type of extract to be produced, more or less hydrocarbon solvent may be left to be recovered by the vacuum oven or used to perform evaporative crystallization. This will be covered in-depth in the isolation chapter.
Certified Jacketed Diamond Miner By BVV
After the desired amount of the hydrocarbon solvent has been recovered, The recovery tank and collection vessels are isolated, and the collection vessel is depressurized after the desired amount of the hydrocarbon solvent has been recovered. The extract is then harvested from the closed-loop system. The residual amount of hydrocarbon solvent remaining within the extract is purged utilizing a vacuum oven, covered in-depth in Chapter 4: Solvent Recovery/Purging. Once the extract has been purged of all residual solvent, it can be further refined through distillation or utilized for infusion in some cases.
1.9CF Neocision Vacuum Oven By BVV
After the closed-loop solvent recovery process has been performed and the extract has been harvested from the closed-loop system the hydrocarbon extraction procedure is complete and the resulting extract can be purged of residual solvent and further refined. Now that we have covered the proper hydrocarbon extraction safety protocols and extraction methodologies let's dive deeper into the extraction preparation and actual operation of a closed-loop system.
Hydrocarbon extraction starts with packing the material column with biomass material and proper assembly of the extractor. During the packing of the material column, botanical biomass is loaded into the material column and packed as evenly as possible to reduce channeling of the solution and tight enough to fit the desired amount of material within the column while still being able to agitate the material by hand. Once the material is loaded into the material column, it is important to remove any debris from the tri-clamp ferrules to ensure a tight seal between the tri-clamp gasket and the endcaps continuing the assembly of the material column.
Packing Rod for Material Tubes By BVV
Once the material column has been packed and assembled, continue to assemble the remainder of the system. While assembling the extractor, ensure that all high-pressure clamps are tightened with a torque wrench to manufacturer specification (typically 110 inches per sqft). After assembly, prior to initiating a hydrocarbon extraction, a pre-assembly inspection and testing of the extractor should always be done to ensure safety during extraction. During the inspection, confirm that all gaskets are free of visible wear and ensure all lines and fittings are intact and appropriately tightened before pressure testing the system.
Glycerine Filled Compound Gauge By BVV
After assembly and preinspection of the closed-loop extractor, it is important to pressure test the system to ensure no leaks. A leak within the system can result in hydrocarbon solvent escaping from the system and being released into the atmosphere. To ensure safety during extraction, it is always recommended to pressure test the system after the reassembly of a closed-loop extraction system. To initiate a pressure test, start by ensuring the outlet to the source solvent tank is closed and then open all process flow valves from the closed source solvent tank to the collection tank and pressurize the system with nitrogen to 100 PSI. Once the system has reached 100 PSI, close the nitrogen input valve and allow the system to be held under pressure for at least 30 minutes prior to extraction to ensure pressure can be held.
BUNA Sanitary Tri-Clamp Screen Gaskets By Rubber Fab
If pressure is not held during this time, identify where pressure leaks are coming from by spraying all process connections with soapy water. If the process connection is leaking, it will be observed to create additional bubbles. Once a pressure leak has been identified, depressurize the system and disassemble and reassemble the system as necessary, making sure to replace any parts no longer holding a seal. Once reassembled, proceed to pressure test the system again, and disassemble and reassemble as needed until pressure can be maintained. At BVV, we pressure test every vessel at 100 PSI for 24 hours prior to shipment upon disassembly and reassembly of the unit. The same is always recommended, but a shorter pressure test can be utilized at the discretion of the extractor.
Once the system has passed the pressure test prepare the hydrocarbon solvent for extraction by bringing it to the desired extraction temperature. If utilizing a new solvent, it is always recommended to distill the solvent to eliminate the risk of contamination from the storage solvent tank before bringing it to the desired extraction temperature.
120LB High Purity Propane tank By BVV
To distill fresh solvent, start by first bonding the source solvent tank to the grounded extractor before injecting hydrocarbon solvent from the source solvent tank into the collection vessel of the pressure tested closed-loop hydrocarbon extractor. Once loaded, close the valve to the solvent storage tank and proceed to heat the collection with a hot water bath to 38C/100F. During this time, it is important to also chill down the recovery tank to condense the ensuing vapors. Once the collection vessel and recovery tank have reached their operating temperature, open all process connection valves between the collection vessel and the recovery tank and proceed to evaporate the solvent from the collection vessel, recondensing it back into the recovery tank. Once all the solvent has been evaporated from the collection vessel and condensed into the recovery tank, close the vapor valve on the recovery tank, depressurize the closed-loop system, and disassemble the collection vessel. Clean the inside of the collection tank with ethanol to remove any residue from the freshly distilled solvent.
Now that the solvent has been distilled and the collection vessel has been cleaned of any residue, proceed to reassemble the system as normal. This will ensure that the system is assembled to manufacture specification, all high-pressure clamps are torqued to specification, and all process connections are secure and appropriately tightened. After assembly, proceed to pressure test the system again for a minimum of 30 minutes to ensure proper assembly before preparing for the initial extraction.
Now that the fresh solvent has been distilled, proceed to bring the solvent to the desired extraction temperature while pulling the remainder of the extraction system under vacuum. Pulling the system under vacuum evacuates ambient pressure from the system, creating a low-pressure zone that allows the hydrocarbon solvent to move through the system more easily and provides a clean atmosphere for the extraction process to be performed. Additionally, pulling an EMPTY recovery tank under vacuum prior to initiating the recovery process creates a low-pressure zone for the solvent vapors to be pushed/pulled towards during recovery.12CFM Two-Stage Spark Free Vacuum Pump By BVV
Before pulling the system under vacuum, start by ensuring the solvent tank is closed off and no residual vapors are within the system. If hydrocarbon vapors are pulled into a standard vacuum pump, it could cause ignition of hydrocarbon vapors. Once the solvent tank has been confirmed to be closed, open all process connections from the closed source solvent tank to the collection vessel. Connect the vacuum line from the collection vacuum valve to an intrensically safe vacuum pump. Ensure all process connections are tightened appropriately and proceed to turn on the vacuum pump and allow the system to be pulled under vacuum to -29.9hg before closing the vacuum valve and turning off the vacuum pump. Once the system has been pulled under vacuum, isolate each component of the extraction system by closing all input and output valves and allowing the hydrocarbon solvent to reach the desired extraction temperature.
Once the extraction solvent reaches the desired temperature, the extraction can be initiated by transferring the solvent from the solvent tank to the material column. If the solvent is warm, the solvent will have its own inherent pressure that can be utilized to inject the solvent into the material column due to the pressure differential being pulled under a vacuum. While this pressure differential is typically sufficient in the transfer from the solvent tank to the material column, if the solvent is chilled enough to reduce the pressure of the hydrocarbon solvent, then nitrogen can be utilized to push the solvent from the recovery tank into the material column of the system. In both cases, nitrogen can push the solvent into the material column by first pressurizing the solvent tank to the desired injection pressure. While higher pressures can be utilized to inject the solvent, 10-25 PSI is typically sufficient in transferring the solvent into the material column.
Explosion Proof Platform Scale By BVV
Once the solvent tank has been pressurized appropriately, place the LP recovery tank on an explosion-proof scale. Tare the scale and open the process connection valves from the source solvent tank to the material column, injecting the desired amount of solvent into the material column, allowing the biomass material to be saturated. If utilizing a quick wash solvent saturation strategy, ensure the process connection valves from the outlet of the material column to the collection vessel are open, allowing the solvent to drain into the collection vessel upon injection. If utilizing a longer saturation, ensure the material column outlet valve is closed, allowing the material to be completely submerged in a solvent for the desired retention time before draining the extraction solution to the collection vessel. Once the entirety of the extraction solution has made its way into the collection vessel, proceed to initiate the solvent recovery process.
To initiate the recovery of solvent within a closed-loop hydrocarbon extractor, start by closing the inlet valve to the collection vessel and then chill down the solvent tank and heat the collection vessel. If the recovery tank is empty, isolate the recovery tank and pull it under vacuum to create a lower pressure zone for vapors to be pulled towards. Depending on the boiling point of the target compounds to be preserved within the botanical extract, more or less heat may be applied to preserve these compounds. Typically the collection vessel will be heated to a temperature range between 20C/68F and 37C/100F to evaporate the hydrocarbon solvent. The application of heat to the collection vessel can be applied by submerging the collection vessel in a hot water bath or recirculating hot water through the jacket of the collection vessel utilizing a heated circulator.7 Liter heated Circulator By Polyscience
Commonly the solvent tank will be chilled as cold as possible by either submerging the recovery tank and condensing coil in a dry ice/ethanol slurry or circulating chilled fluid through the jacket of the solvent tank utilizing a refrigerated circulator. Once both the collection vessel and the recovery tank have reached the desired temperature, initiate the solvent recovery process by opening all process connection valves between the collection vessel and the recovery tank. If utilizing an active recovery pump, turn it on at this time. During this time, closely monitor the extract within the collection vessel to gauge the solvent recovery process. The solvent recovery process can be continued until the extract completely solidifies. No more boiling is observed, requiring it to be scraped out of the collection vessel. It can also be stopped once only a small amount of the extraction solvent remains to allow the extract to be poured or drained from the collection vessel and purging the remainder of the solvent utilizing a vacuum oven.
Once the solvent recovery process has been performed to the extractors liking, isolate the solvent tank and collection vessel, turn off the recovery pump if utilized, depressurize the closed-loop extractor, and harvest the extract. Once the extract has been harvested, proceed to the vacuum purging procedure outlined In Chapter 4: Solvent recovery/Purging.

HYDROCARBON EXTRACTION STANDARD OPERATING PROCEDURE
Purpose
The purpose of this procedure is to provide detailed instructions for closed-loop hydrocarbon extraction.
Scope
This procedure applies to all lab technicians tasked with closed-loop hydrocarbon extraction.
Definitions/Acronyms
Personal Protection Equipment (PPE) Items worn to protect employees from exposure to hazardous materials and prevention of injury.
Safety Data Sheet (SDS) Provides useful information on chemicals, describing the hazards the chemical presents, and giving information on handling, storage, and emergency measures in case of an accident.
Safety
SDS Sheets: Butane SDS, Propane SDS, Tri-Blend SDS
PPE: The following should be worn by all lab personnel during this refinement procedure:
Protective eyewear
Lab coat/Anti-static clothing
Nitrile Gloves
Respirator
Preparation and Use:
Target compounds will be extracted from biomass material utilizing hydrocarbon solvent.
Quantity- 3-5lb of hydrocarbon solvent per 1lb of biomass
Frequency- An initial volume of solvent is used, no more is added.
Location- Hydrocarbon extraction occurs within a C1D1 environment, inside of a closed-loop hydrocarbon extractor (BVV).
Potential Hazards and Risks
See hydrocarbon solvent SDS for detailed risks.
6. Preparation
7. Procedure
Disassembly and cleaning




